Regulatory guidelines increasingly require pre-use post-sterilization integrity testing (PUPSIT) for sterilizing-grade filters. Manufacturers want to get it right—that’s why we’re here to lend a helping hand.
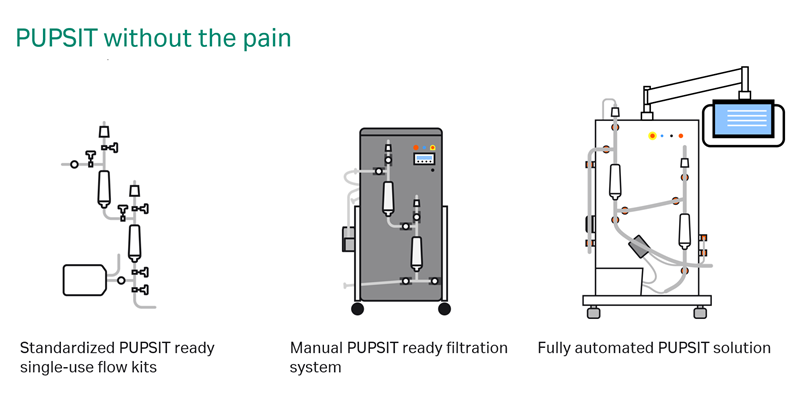
Whether you’re looking to implement or improve your PUPSIT operations, we’re here to guide you every step of the way. No matter where you are in your journey, Cytiva offers a range of products and services—including manual and automated solutions for performing PUPSIT, and validation and regulatory support to ensure compliance.
Ready to take the next step? Let’s navigate the path to meeting your PUPSIT needs together.
Fill out the form to find your PUPSIT solution today.
Complete the form to find your PUPSIT solution
*required fields
Acrodisc® Syringe Filters
Reliable Results with Superior Protection
Form title
Form sub title
Panel 2 header
Panel 3 header

Follow our connected mAb perfusion process
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .
Curious about continuous biomanufacturing? Join the Cytiva project team vlog series as we document our proof of concept trial and provide insights to help you efficiently plan and execute your project .


