Simplify and scale
your gene therapy manufacturing
Consistent, high-quality AAV production
with the ELEVECTA™ producer cell line
Fill out the form to request a meeting or ask a question
Here’s how we work together to elevate your AAV production to the next level
We engineer a stable producer cell line with your capsid and GOI. A research master cell vial is handed over to you to create your good manufacturing practices (GMP) cell bank. Then just expand, scale up, and induce from a single cryovial to start your AAV production.
See how the ELEVECTA™ producer cell line’s one-step process ensures robustness and scalability for your gene therapy endeavors.

Stable, inducible AAV production
ELEVECTA™ producer cell line
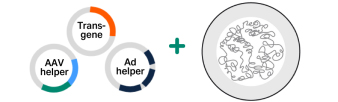
The HEK293-based ELEVECTA™ producer cell line offers a solution for stable and inducible adeno-associated virus (AAV) production. With all required genes stably integrated into the host cell's genome, AAV production is initiated with a simple induction step, eliminating the need for multi-step transfection.
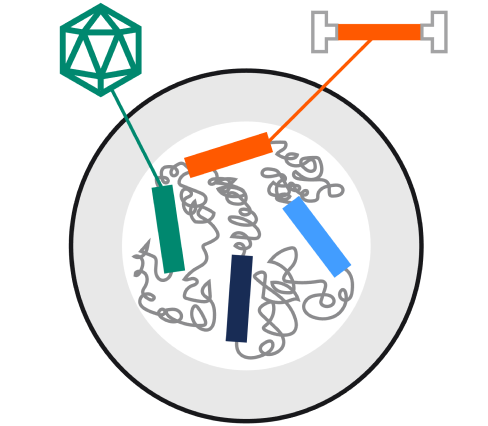
Integrated genes for streamlined production
With Rep, Helper, Cap, and the gene of interest (GOI) stably integrated into the host cell’s genome, there's no need for plasmid transfection or helper virus. This integration simplifies the production process and enhances efficiency.
Benefits of the ELEVECTA™ producer cell line
- Simplified manufacturing process: Streamline your AAV production process by replacing multi-step transfection with a single-step production method. Just add the induction agent, and you're ready to go.
- Easy scalability: Achieve consistent and high-quality whether you're producing small batches or scaling up for large quantities.
- Enhanced reproducibility: Consistency is key! The monoclonal nature of the cell line guarantees that AAV is produced identically in every batch, ensuring reproducibility and reliability.
- Reduced cost of goods: By eliminating the need for expensive transfection reagents and plasmid DNA, you can significantly reduce your cost of goods.