ADC Integrated Solutions: Streamline manufacturing for lower risk
Would you like to learn more? Talk to our specialists to discover what’s possible.
Please read our Terms and Conditions and simply fill out the contact form below.
Advance ADC therapies faster with a fully integrated solution
Bringing antibody-drug conjugates to market is complex – and multi-vendor models often lead to delays, data gaps and inconsistent quality. Our ADC integrated solution unifies every step ensuring a seamless coordination with strong quality control to accelerate development. With everything designed to sync together, you get a faster, more reliable path to breakthrough cancer treatments.

Have you asked yourself why having an integrated ADC solution matters?
Antibody-drug conjugate development is a tightly interconnected process – every decision in antibody design, linker-payload selection, conjugation and purification strategy impacts the next. When these processes are split across multiple vendors, data becomes fragmented, timelines stretch, and quality can vary from partner to partner. An integrated ADC solution removes these barriers by keeping an entire workflow under one coordinated process.
- Unified workflow: Eliminates the communication gaps and handoff issues common with a multi-vendor setup
- Consistent quality: Shared standards, methods and controls reduce variability and improve reliability
- Stronger data continuity: Centralized information improves traceability, analysis and regulatory readiness
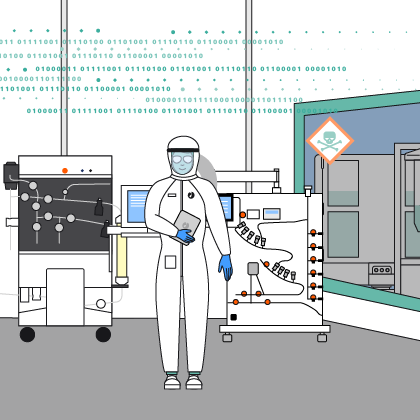
- Increase safety: Reduce cleanroom entries and potential operator exposure to cytotoxic material
- Smoother scale-up: High quality early-stage work transitions naturally into full GMP manufacturing
We offer a broad portfolio of learning and training solutions designed to strengthen skills, close capability gaps and build confidence across bioprocessing teams.
We offer flexible programs that meet your needs.
Fast Trak™ training: Instructor lead courses with hands-on training provides education across upstream, downstream and manufacturing operations.
Digital learning: Interactive, on-demand eLearning and virtual modules support blended learning strategies.
In-person training: These sessions provide hands-on experience with our equipment, real-world process simulation and guidance from subject matter experts.
We provide comprehensive equipment services designed to keep your systems performing reliably from installation through end of life.
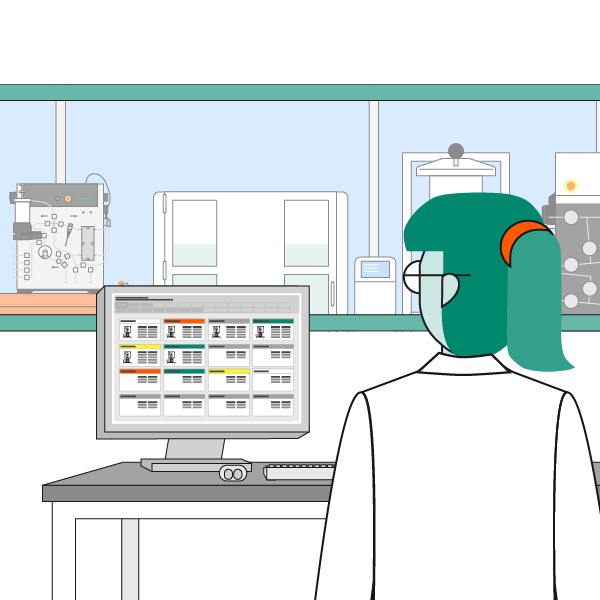
Smarter Control. Unified insights. Confident operations.
- A unified monitoring and control layer that consolidates equipment, assets and distribution infrastructure
- Real-time operational awareness with alerts, diagnostics and performance insights in one place
- Actionable intelligence that helps reduce downtime streamline processes and elevates reliability
- Flexible integration
- Intuitive data-rich dashboards
- Robust security frameworks
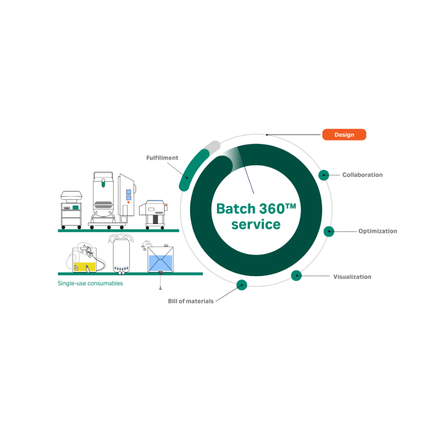
Batch360™ single-use consumables deliver plug-and-play reliability across upstream, downstream, and fill-finish workflows – engineered to reduce changeover time, minimize contamination risk, and standardize performance from lab to GMP manufacturing.
Batch360 single-use technology brings clarity and control to ADC development. By uniting digital traceability with standardized assemblies, it reduces material variability, streamlines setup and lowers the risk of contamination.

Bring E&L data into a unified digital environment – gain a central source of truth. Robust extractable and leachable data is essential for accelerating ADC development.